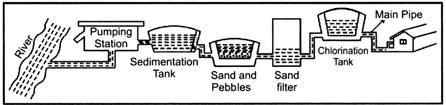
States of Matter
- We see a variety of objects around us. Generally, all objects are classified into three categories:
(i) Solids (stone, wood, iron, etc.)
(ii) Liquids (water, oil, milk, etc.)
(iii) Gases (oxygen, air, vapour, etc.)
- Water can exist in all the three forms—solid, liquid, gas.
- Solids and liquids have surfaces but gases do not have surfaces.
- Liquids need a container to kept it, otherwise they tend to flow. On the other hand solids can be heaped anywhere.
- All matters are made up of tiny particles called molecules. Molecules are constantly moving about. The higher the temperature, the greater is their motion. Also, when two molecules come closer they tend to stick together because there is a weak force of attraction between them.
- The movement of molecules in solids, liquids and gases has been described:
Solids: In a solid, the intermolecular forces are so strong that molecules cannot slip away. They are kept together in order and are not allowed to move much.
This makes it clear why solids melt on heating and liquids turn into gases when heated. Also, when a gas is cooled, its molecules slow down and eventually condense into a liquid.
Liquids: In a liquid, the molecular motions are not great enough to overcome the force of attraction between the molecules. This is why, the molecules tend to stay together and give a condensed form. However, these forces are not strong enough to prevent the liquid from flowing.
Gases: In a gas, molecules are moving fast. They overcome the molecular force of attraction and move in all directions. This is why, a gas keeps expanding until it fills all the available space.